We occasionally receive excellent questions and/or comments by email or via our contact form and have then usually corresponded with the emailer directly. But, some of the questions and answers deserve a broader audience, so we decided to highlight some of them in a new series of blog posts.
For the average citizen, who perhaps has a rudimentary grasp of general science, one problem with the topic of climate science is that it is such a huge subject which draws on a wealth of knowledge from many different science disciplines. It is easy to get lost in the weeds of this detailed knowledge which informs our conclusion that the current changing climate is due to human emissions of greenhouse gases. The broad strokes of climate science may be grasped with a limited amount of scientific knowledge: carbon dioxide and other greenhouse gases absorb and release heat (infrared radiation)...burning fossil fuels releases CO2 into the atmosphere and this increasing amount can be measured...more CO2 in the atmosphere will mean more heat energy in the climate system causing climate to change. But that basic knowledge is grounded on many more complicated lines of evidence: the weeds, which can often be very confusing to the science novice.
Recently, our inbox has seen a fair number of emails from folks asking about carbon isotopes. Some emailers asked basic questions while others asked about some finer details of the more complicated science. This post will offer a primer on some of the basics about what isotopes are, where they come from, and what isotopes can tell us about Earth's climate. In a second post we will get into the weeds a bit and explain how carbon isotopes are measured.
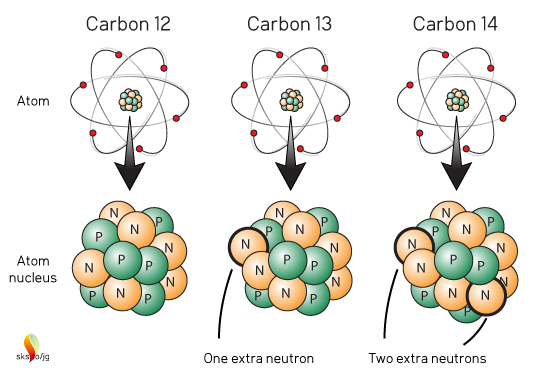
All atoms are made up of protons (positive charge), neutrons (no charge) and electrons (negative charge). The number of protons in an atom's nucleus defines what element that atom is. For the element carbon, all atoms have 6 protons and 6 electrons (Figure 1). Most carbon atoms also have 6 neutrons, hence 6 protons + 6 neutrons = carbon-12 or 12C. But a small number of carbon atoms have an extra neutron, carbon-13, or 13C and an even smaller number of carbon atoms have two extra neutrons: carbon-14, or 14C.

Figure 1. Structure of the three naturally occurring carbon isotopes.
These three, naturally occurring "flavors" of carbon atoms are called Isotopes ("same type"). Chemically, they all act like carbon atoms, they all "look" like carbon atoms. The 13C and 14C atoms are just slightly heavier than 12C. The vast majority of carbon on Earth is 12C with about 99%. 13C is about 1%, and 14C much, much, much less than 1% (about 1 atom of 14C per 1 trillion carbon atoms). Think of those ratios as the baseline or background ratios of most naturally occurring carbon on Earth. Take a random rock, or block of wood, or pound of flesh and measure the carbon in it and the ratios will be very close to 99%:1%:<<<1%. Well almost, but we'll get to that later.
12C and 13C are "stable" isotopes which do not change, but 14C is "radioactive" (often called "radiocarbon") and decays over time into nitrogen. The "half-life" for 14C is 5730 years, which means that if there are 100 atoms of 14C in some sample, 50 of them will decay to nitrogen in 5730 years. In another 5730 years, 25 of those remaining 50 14C atoms will decay, etc. Eventually, virtually all of the 14C atoms in the sample will decay to nitrogen. And nitrogen in Earth's atmosphere is where 14C atoms originate.
14C is formed in the atmosphere by cosmic rays (Figure 2). These "rays" are actually high-energy particles rather than light rays (electromagnetic radiation), although they move at nearly the speed of light. Most cosmic rays are the atomic nuclei of hydrogen (a proton) or helium (two protons and two neutrons, also called an alpha particle) which have been stripped of their electrons. They come zipping into the atmosphere from outer space and undergo various transformations which generate neutrons. These neutrons then collide with nitrogen nuclei (the most abundant element in the atmosphere) converting them to 14C and an extra proton which the new 14C spits out.
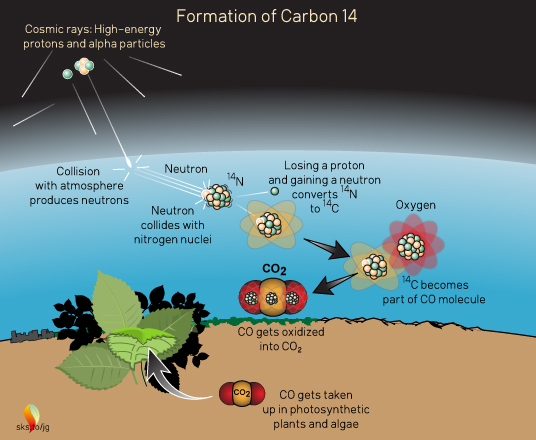
Figure 2. Formation of carbon-14 in the Earth's atmosphere.
These high-energy reactions leave the individual 14C atoms with a lot of kinetic energy, causing them to crash into many other atoms/molecules in the air. They zip about grabbing electrons (which get stripped off in nuclear reactions) and eventually join with oxygen to form CO which can then be oxidized into CO2.
Since all carbon isotopes are the same chemically they can all interact with other atoms and molecules in the same way. In photosynthesis, living plants take in CO2 from the air, energy from the sun, and water from the ground to make the building blocks of living matter and the energy they need to grow. All three carbon isotopes show up in the CO2 which plants use in photosynthesis, however, since 13C and 14C are slightly heavier than 12C, the plants "prefer" to use the lighter isotopes of carbon: 12C over 13C over 14C. So if you took a plant and measured what isotopes it has you would find that it would be slightly "richer" in 12C than in the naturally occurring background ratios. So it might have 99.2% 12C, 0.8% 13C, and much, much less than 1% 14C. And since animals eat plants (and other animals which eat plants) their isotopic ratios would be similar to plants.
Now, imagine what happens to plants which die and get buried in the muck of a swamp somewhere. They don't really decay and no critters eat them, they just get buried deeper and deeper by muck and other plant material. Eventually, given enough time, virtually all of the 14C will convert to nitrogen. If we dig them up some millions of years later and measure their isotopes they will just have 12C and 13C.
This is exactly how we get fossil fuels: tons and tons of plant material get buried and compressed over vast stretches of geologic time. By the time we dig them up any 14C that was there has long since decayed. So when we burn these fuels the carbon we are adding to the atmosphere is enriched in 12C and 13C, but depleted of 14C. And, since the fossil fuels come from plant material, the carbon is also richer in 12C versus 13C. So now when we measure the atmosphere's CO2 we can see that over the last hundred years or so the natural background ratios of 12C:13C:14C are getting more and more skewed to more 12C, and less 13C and 14C. This is one of the many lines-of-evidence which show that the rise in CO2 in the atmosphere is caused by the burning of fossil fuels.
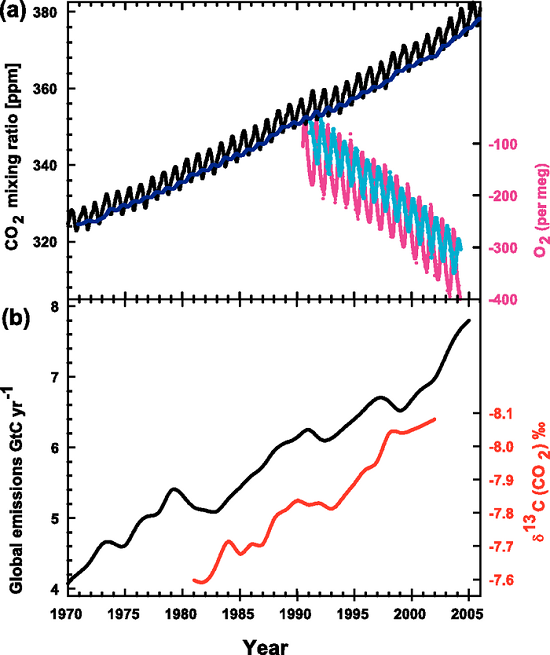
Figure 3. Recent CO2 concentrations and emissions. (a) CO2 concentrations over the period 1970 to 2005 from Mauna Loa, Hawaii (black) and Baring Head, New Zealand (blue). In the lower right of the panel, atmospheric oxygen (O2) measurements from flask samples are shown from Alert, Canada (pink) and Cape Grim, Australia (cyan). (b) Annual global CO2 emissions from fossil fuel burning and cement manufacture in GtC yr–1 (black) through 2005. Annual averages of the 13C/12C ratio measured in atmospheric CO2 at Mauna Loa from 1981 to 2002 (red) are also shown. The isotope data are expressed as δ13C(CO2) ‰ (per mil) deviation from a calibration standard. Note that this scale is inverted to improve clarity. (IPCC, AR4)
The bottom panel (b) of Figure 3, from the 4th Assessment Report of the IPCC, illustrates this line-of-evidence. The black line shows the increase in CO2 emissions from fossil fuel burning, and the red line shows the declining ratio of 13C to 12C in the atmosphere's CO2. (It may look like the ratio is increasing along with our emissions but notice that the scale for the red line is reversed.)
The top panel (a) of the graph shows the familiar rise in atmospheric CO2, measured in parts per million (ppm): the black, saw-toothed line. The carbon isotopes are measured quite differently, in δ13C (CO2)‰. In Part 2, I will explain what this strange incantation means.
Thanks to Sarah, Dikran, and jg for help with this post.
Posted by David Kirtley on Wednesday, 20 December, 2017
 |
The Skeptical Science website by Skeptical Science is licensed under a Creative Commons Attribution 3.0 Unported License. |