This is an update to a previous analogy. The original version is here.
Greenhouse gases (GHGs) determine amount of warming, but oceans delay the warming.
Try this thought experiment.
To see how water delays the warming, try this thought experiment.
How much longer does it take to reach 60°C (about 140°F) with water instead of air in the pot? A lot longer!
The longer time required to heat a pot of water than a pot of air explains why there is a delay between GHG emissions and a rise in temperature of the atmosphere: the oceans take a long time to warm up. This is why scientists, such as James Hansen, refer to global warming as an inter-generational issue, because the time lag means that the heating due to our emissions are only fully felt by later generations.
The earth is covered mostly by water. The large heat capacity of the oceans soak up a lot of energy and slow down the heating of the atmosphere. Just how long is the delay between the time we inject CO2 and other GHG's into the atmosphere and when the effect is felt?
For a given CO2 concentration, given enough time and keeping the CO2 concentration constant, we can estimate the final temperature the atmosphere will reach by using the average IPCC estimate of 3°C warming for doubling CO2 concentration (called the “equilibrium climate sensitivity”, ECS). Using the estimate of pre-industrial CO2 concentration of 280 ppm (parts per million), a climate sensitivity of 3°C implies that CO2 concentrations of 350, 450, and 550 ppm yield 1, 2, and 3°C warming, respectively. Using this estimate of climate sensitivity, together with measurements of CO2 from 1970 to today, we can estimate the warming that corresponds to current atmospheric CO2 concentrations. That is, knowing the "burner setting", we can estimate the final temperature of the pot of water, even though we will have to wait some time for it to heat up.
I use the GISS land-ocean data (Goddard Institute for Space Studies) to plot measured global mean temperature above pre-industrial, to estimate the time lag between the temperature anomaly suggested by a particular CO2 concentration and the time when that temperature is observed. This and CO2 concentrations for five selected years are shown in the following figure.
Figure 1. Expected warming based on atmospheric CO2 concentration, using an ECS = 3ºC/doubling CO2, vs committed warming based on measured temperature anomalies (GISS land-ocean data).
Figure 1 indicates 2 things: (1) the time lag between emitting greenhouse gases and when we see the principal effect is about 30 years, due mostly to the time required to heat the oceans, and (2) the rate of temperature increase predicted by a climate sensitivity of 3°C tracks well with the observed rate of temperature increase. As of 2022 we have reached 420 ppm CO2, corresponding to 1.7ºC, if CO2 concentrations remain at current levels.
So whereas the experiment at home with a pot of water on low heat yields a time lag of something like 10’s of minutes to heat the water, to heat an Earth-sized pot of water with GHG's increasing at current rates, the time lag is 10's of years.
Notice in that last sentence I said "with GHG's increasing at current rates." With all of the talk of net-zero emissions, or perhaps just accepting stabilizing CO2 concentration as an acceptable, interim goal, what would happen to the warming and the time lags if we were to stabilize CO2 concentration at 2022 levels? Let's try that thought experiment.
Zeke Hausfather (read here) reports that to stabilize CO2 at current levels, we would need to reduce CO2 emissions by about 70%. To keep CO2 at that level, we would need to continually reduce emissions until we reach net-zero emissions in about 100 years. So the point is that stabilizing CO2 concentration is only an interim goal. Long term, to keep CO2 from rising, we must reach net-zero emissions at some point. Figure 2 shows the hypothetical situation where CO2 is stabilized at 2022 levels for about 100 years, until it reaches net-zero emissions at that point. Think of the temperature difference between committed and equilibrium temperatures as a spring, pulling the committed temperature up to the equilibrium temperature. The oceans resist the action of the spring. Because we have dramatically reduced CO2 emissions from current rates in our thought experiment, instead of the ocean time lag being several decades, it is more like a century.
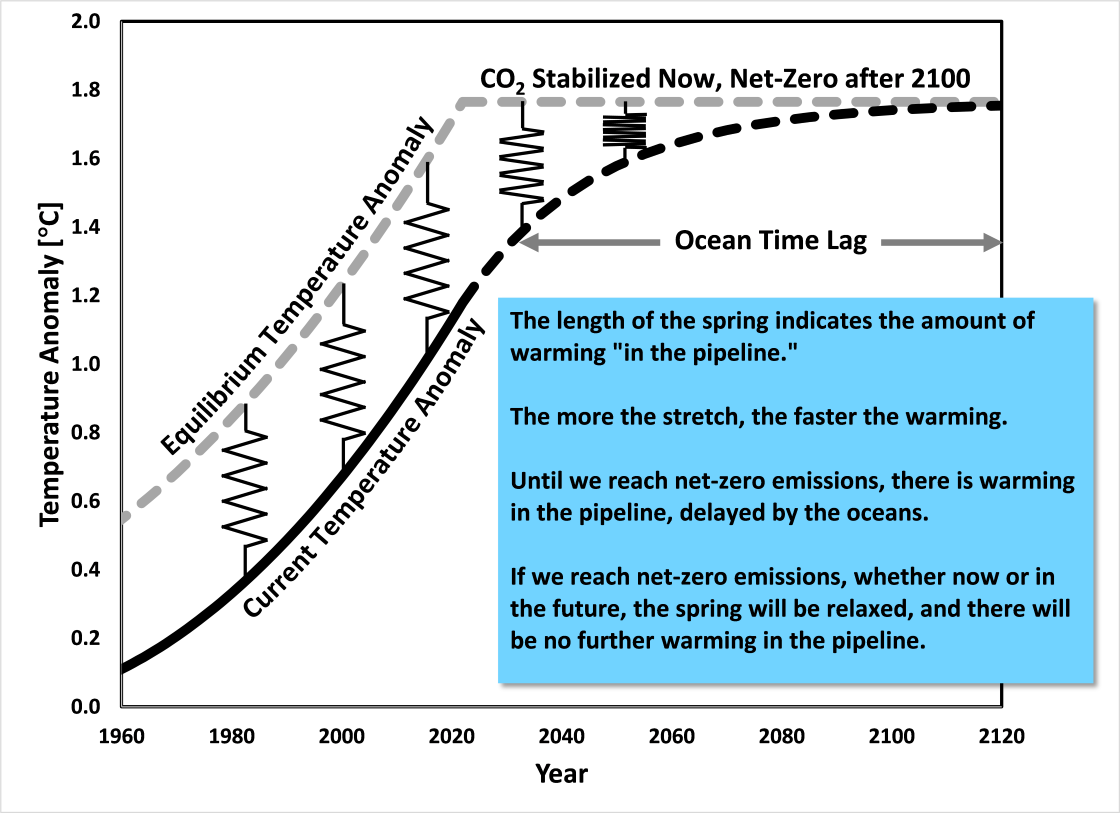
Figure 2. Temperature trajectory assuming atmospheric CO2 stabilization near 2022. To achieve this trajectory, CO2 emissions must first drop about 70% and then continually decrease until they reach net-zero emissions about 100 years later. About 100 years are required for the temperature to stabilize because of the time required to warm the oceans.
Long story short, the oceans are helping us out a lot by slowing down the warming. Of course, once we warm the earth, the oceans will also slow down the rate of cooling, should we ever try a global-cooling experiment. The ocean time lag works both ways.
Posted by Evan on Monday, 4 July, 2022
 |
The Skeptical Science website by Skeptical Science is licensed under a Creative Commons Attribution 3.0 Unported License. |