The decline of the Great Barrier Reef
What the science says...
The evidence clearly shows that ocean warming and acidification due to human CO2 emissions are adversely impacting the Great Barrier Reef.
Climate Myth...
Great Barrier Reef is in good shape
the Great Barrier Reef is in fine fettle (Bob Carter)
What's the current state of the GBR (i.e. is it really "in fine fettle")?
Despite being one of the best managed marine ecosystems worldwide, there is evidence that the ecological 'health' of the Great Barrier Reef has declined since the arrival of European settlers into the Queensland region. This evidence comes from a number of key sources. This area is not without its controversy, which is discussed elsewhere at Skeptical Science by Professor John Bruno (University of North Carolina) and others.
Historic photographic analysis (> late 19th century)
One of the first and most direct way has been via 'before and after' photographs assembled by Dr. David Wachenfeld of the Great Barrier Reef Marine Park Authority. Dr. Wachenfeld searched photographic archives of pictures of intertidal coral reef systems going back over 100 years. He then organised photographs to be taken in exactly the same spot of the same reef systems. The results revealed no change in reefs systems at 6 out of the 14 sites where it was possible to compare photographs from 100 or more years ago. At four sites, there was evidence of change in the health of intertidal reef communities, while at the 4 remaining sites, coral had more or less disappeared from the intertidal reef areas (Wachenfeld 1997). Photographic analysis is a very direct way of assessing whether or not coral reefs have declined at particular locations. However, it is important to acknowledge that photographers probably went out to photograph 'beautiful' areas of the reef as opposed to areas without coral cover. The criticism that arises is that had that happen, we might have seen an equal and opposite set of photographs in which coral returns to coastal reef flats. Several more recent studies have been exploring sediments using push coring and radioisotope dating techniques, which is allowing a reconstruction of past communities based on the sediments that have built-up. These studies are revealing that major changes have been occurring in coastal coral communities, with the general trend that coral communities have converted into seaweed communities that are very different to the coral communities of old.

Figure 1. Matched photographs of the same section of reef taken around 1890 by William Saville- Kent and again in 1994 by Andrew Elliott. Full details can be obtained in the study reported by Wachenfeld (1997).
Meta-studies: long-term trends (> 1960)
Several research groups have assembled datasets from the growing number of studies that have been undertaken on the Great Barrier Reef. These studies are referred to as meta-studies given that they depend on drawing numbers from published papers. The idea is that if these sources are carefully vetted for a certain level of quality and rigor, then the dataset of numbers on things like the abundance of coral on coral reefs at a certain time in recent history can be established. One of the first of these was (Bellwood et al. 2004) who found a strong negative trend in coral abundance on Great Barrier Reef sites going back to 1960. This analysis supported a related study by (Pandolfi et al. 2003) who used historic and paleontological evidence to compare the Great Barrier Reef to other reef systems worldwide. They also concluded that the Great Barrier Reef, although still a beautiful reef system had declined over the past century. The third study by (Bruno and Selig 2007) undertook a similar analysis, and compared coral cover on the Great Barrier Reef in the early 1980s to recent survey data. This study revealed a major drop in coral cover (40-50%).
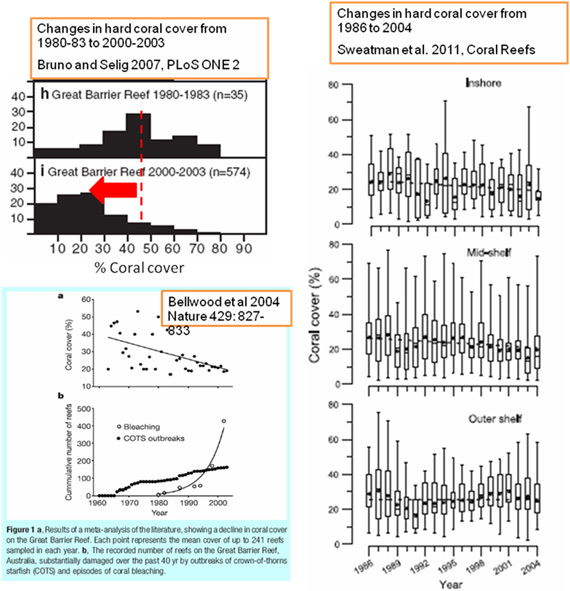
Figure 2. Three independent studies reporting changes in the percentage of coral cover on reefs within the Great Barrier Reef Marine Park.
AIMS long-term ecological surveys (>1986)
The third category of information comes from long-term survey data. The Australian Institute of Marine Science began annual ecological survey of key aspects of coral and fish abundance on the Great Barrier Reef in 1986. While these surveys biased toward offshore reef areas and hence have probably not caught the full picture of human impacts on the Great Barrier Reef, they are empirically sound in terms of the methodology needed to examine ecological parameters such as coral abundance over time. As indicated above, there has been some scientific debate (which always goes on and is part of the sceptical process of science) and some of the details have been discussed by Professor John Bruno in separate postings to SkS and Climate Shifts.
Drawing together these three types of studies, there is fairly compelling evidence that the Great Barrier Reef has undergone significant ecological change over the past 50 to 100 years. Even the declined reported by the AIMS Long-Term Monitoring Project (Figure 2) from 28% to 22% coral cover (a decrease of 22%) from 1986 to 2004 (Sweatman et al. 2011) is of great concern given the massive size of the Great Barrier Reef and the speed of this change (10% decrease per decade).
How do we know the GBR decline is due to human impacts?
Evidence for humans being responsible for the recent changes in the Great Barrier Reef comes from a number of different disciplines and sources.
Firstly, there is a considerable body of information within the literature that has examined the sensitivity of reef building corals, a core component of coral reefs, to stressors such as high temperatures and light levels, reduced salinity and alkalinity, elevated nutrient loads, sedimentation, toxins, and pollutants. This information has been assembled in a number of different review articles and can be accessed through scientific journals such as Coral Reefs. The peer-reviewed science in these journal articles deal with the core sensitivity of reef building corals to particular factors.
Secondly, there is abundant field evidence of these types of factors having a big impact on reef ecosystems. In this regard, there are numerous examples of sudden changes to environmental parameters (e.g. flood damage through reduced salinity to restructures, mass coral bleaching events triggered by elevated sea temperatures) which are consistent with the information from more laboratory and aquarium-based studies. Taken together, the two are constantly interacting. Observations in the laboratory that are not supported by field evidence generally trigger reinvestigation in the laboratory, and vice verse.
Lastly, there are modelling studies which summarise our understanding of how coral reefs might react to changes in the physical, chemical, and biological changes within the environment. These studies have provided projections or predictions which can be tested. In cases where a projection or prediction fails to be supported by laboratory and field evidence, models are then re-examined and reconfigured. The most important step, however, is to ensure that models that have been reconfigured are subsequently tested. Without this final step, one can fall into the trap of models that are adjusted to fit the results as opposed to being truly predictive in nature.
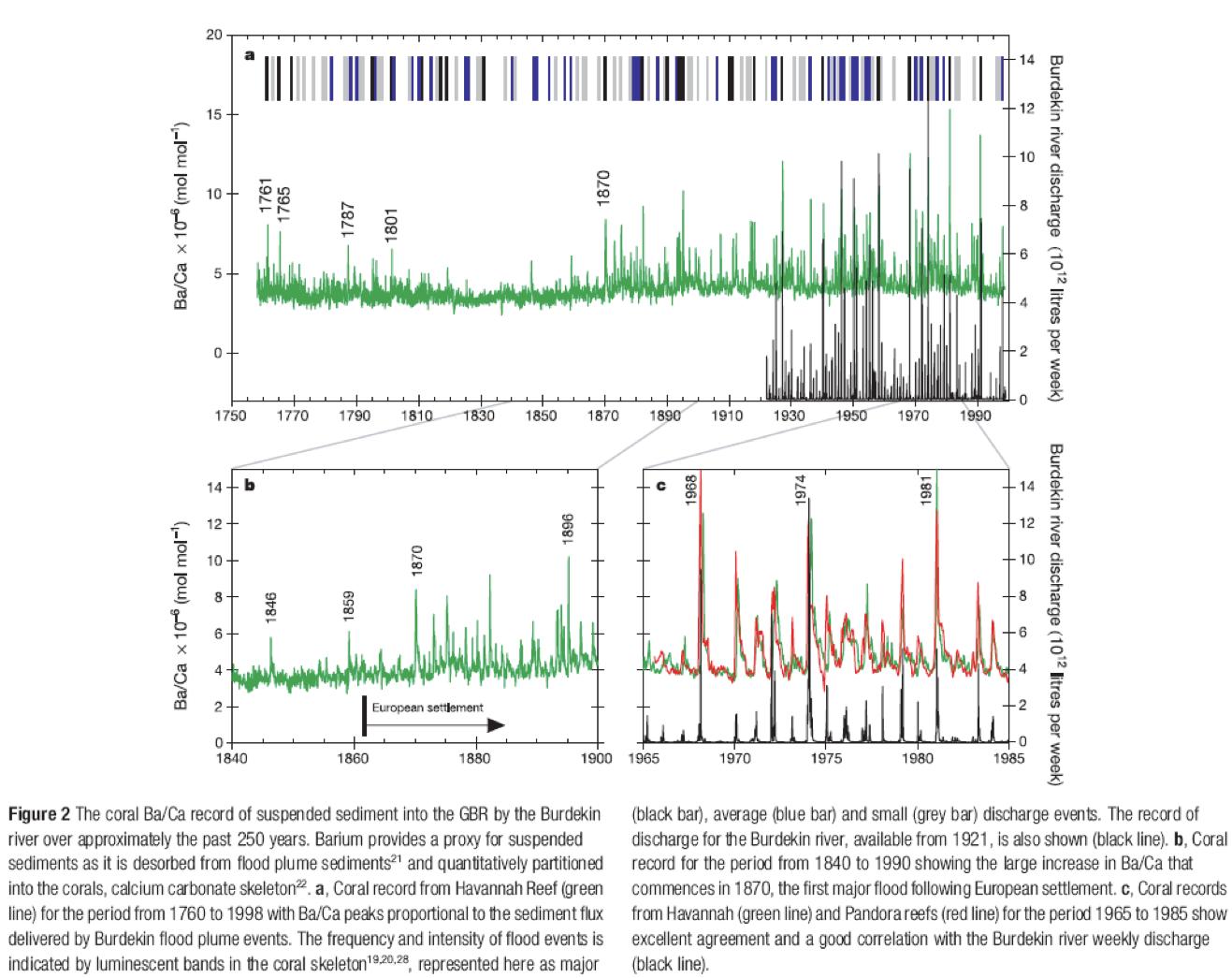
With these datasets in place, how are we able to link human activities to the changes that we are seeing? This comes down to the link between the stressors (elevated temperatures and light levels, reduce salinity and alkalinity, elevated nutrient loads, sedimentation, toxins and pollutants) and human activities. At local levels, there are extremely strong links between changes to land-use along the Queensland coast and elevated levels of sediments, nutrients, toxins and pollutants, and low salinity events. As trees were cut down in the major river catchments, for example, soil was destabilised along the banks of the rivers and creeks that feed into the coastal areas of the Great Barrier Reef. This led to much higher levels of these known stressors. Interestingly, there have been some studies (McCulloch et al. 2003) that have used isotopic information to show that the levels of these stressors have increased by 5-10 fold since the modification by European farmers of river catchments within the Great Barrier Reef basin.
From McCulloch et al. (2003)
These types of data have built up a comprehensive set of linkages between human activities such as deforestation, intensive agriculture, and fishing. There are a number of resources that are available at the Great Barrier Reef Marine Park Authority and at the Australian Research Council Centre for Excellence in Coral Reef Studies and explore the evidence and background to our concern about these local factors.
Is climate change a big threat to the Great Barrier Reef and how do we know?
Coral reefs like the Great Barrier Reef depend on a narrow set of environmental conditions within which they prosper. At the heart of their biology, is a symbiosis that they form with tiny plant-like organisms known as dinoflagellates (commonly called zooxanthellae). This symbiosis is critical to the survival of corals and coral reefs, making possible the efficient trapping of sunlight by reef-building corals. This allows them a cheap source of energy with which to grow in the sunlit waters along tropical coastlines, and where they often deposit of vast quantities of calcium carbonate (a limestone-like substance), creating the three-dimensional structures that we know of as coral reefs. These magnificent structures build up over time, attracting over one million species which are known to live in and around coral reefs (Reaka-Kudla 1997). This mega-diversity is unrivalled anywhere else in the ocean, and supports industries like tourism and fisheries, and provides food for at least 100 million people worldwide (Hoegh-Guldberg 1999).
Coral reefs have come under increasing pressure from the local effects discussed in Part 1 which arise from the rapidly growing coastal populations worldwide. In the early 1980s, coral reefs all over the world began to exhibit a phenomenon known as mass coral bleaching. Mass coral bleaching occurs when the symbiosis between corals and tiny plant-like organisms known as dinoflagellates breaks down. As a result, corals are deprived of their energy source, leaving them prone to disease and death. Over the past 25 years, mass coral bleaching and mortality events have grown in frequency and scale.

Figure 4. Background image shows and extensively bleached reef near Great Keppel Island on the southern Great Barrier Reef in early 2006. The insert shows a normal looking coral on the left-hand side and a bleached coral on the right hand side. The dinoflagellates symbionts which leave the coral is it bleaches are also shown.
The observation that there are no reports of mass coral bleaching prior to 1980 in the scientific literature has led many to conclude that mass coral bleaching is a new phenomenon associated with the activities of humans. The flipside to this argument is that mass coral bleaching events may have occurred in the past but we've only just begun to notice them. This argument falls down for a number of reasons. Firstly, mass coral bleaching events are hard not to notice. When a reef undergoes mass coral bleaching, there is a brilliant white colour that can be seen from passing satellites. Secondly, these events cover hundreds of square kilometres of territory when they occur, and would be as hard not to notice as a bushfire would be on land. Given that scientists have been intensely studying coral reefs since the 1950s (as verified by the scientific literature which contains thousands of articles on coral reefs prior to 1980), it is inconceivable that observers would have failed to note this massive visual change in the ecosystem that they were studying. Lastly, filmmakers such as Ron and Valerie Taylor who have been filming coral reefs since the 1960s do not have mass coral bleaching images on film.
The Great Barrier Reef has experienced six major bleaching events since they began occurring. These events have been growing in scale and size, culminating in 1998 and 2002 in mass bleaching events which affected over 50% of reefs within the Great Barrier Reef Marine Park (Berkelmans; Oliver 1999; Berkelmans et al. 2004).
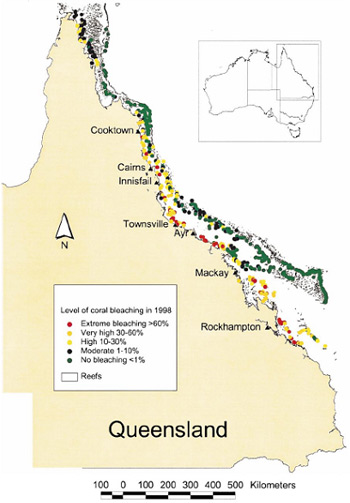
Figure 5. Geographic patterns associated with mass coral bleaching in 1998 (Berkelmans; Oliver 1999).
There is little doubt within the scientific community that elevated sea temperatures are driving the increase in mass coral bleaching and mortality seen on the Great Barrier Reef over the past 25 years. The evidence comes down to 3 key components:
Firstly, experimental studies within aquaria and laboratory settings revealed that corals will bleach when exposed to increases in sea temperature of around 2 to 3°C above their normal maximum temperatures (Glynn; Dcroz 1990; Hoegh-Guldberg; Smith 1989; Jones et al. 1998).
Secondly field study measurements have revealed a close association between small changes in sea temperature above long-term summer maxima and mass coral bleaching events in the field (Berkelmans 2002; Bruno et al. 2001; Dove; Hoegh-Guldberg 2006; Glynn 1983, 1993, 1996; Oliver et al. 2009).
Lastly, satellite detection algorithms based on the laboratory and field evidence have been highly successful in predicting the incidence of mass coral bleaching based on small changes in sea surface temperature. In this case, satellites flown by the National Oceanic and Atmospheric Administration as part of their Coral Reef Watch Program are highly accurate in terms of predicting when and where bleaching will occur.
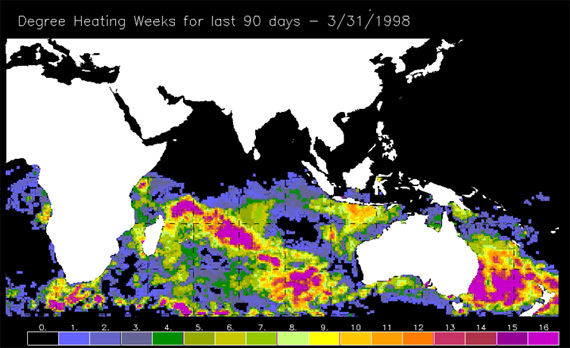
Figure 6. Exposure to elevated heat stress during the lead up to the 1998 bleaching event. Degree heating weeks are calculated by multiplying the size of the sea surface temperature anomaly (relative to the long-term summer average) by the time of exposure. Anything over 4 results in bleaching, while anything above 8 is likely to result in large-scale coral mortality.
Mass coral bleaching represents a threat to coral reefs everywhere. Seawater temperatures are increasing in tropical waters at the rate of 0.1-0.4 degrees Celsius per decade, and will soon be high enough to cause mass coral bleaching every year. Several analyses have been done for coral reefs across the planet (Done et al. 2003; Donner et al. 2005; Hoegh-Guldberg 1999) which have used the known sensitivity of coral reefs and projections of how sea temperature will change over the coming decades. These studies also revealed that coral reefs will bleaching conditions on an annual basis as early as 2030. Given the mortality rates of corals following mass coral bleaching events can be extremely high (over 90% of all corals on a reef dying within a single mass bleaching event) and the fact that it takes at least 15-30 years for a coral reef to recover from a mass mortality event, these trends strongly suggest that coral-dominated ecosystems are very likely to disappear by the middle of the century.
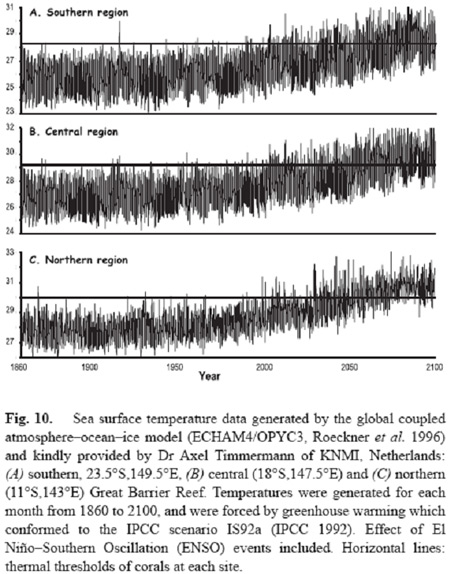
Figure 7. Projected sea temperature projected for the southern, central and northern Great Barrier Reef using a global circulation model (ECHAM4/OPYC3). The horizontal lines in each figure represents the temperature thresholds above which significant bleaching and mortality occur.
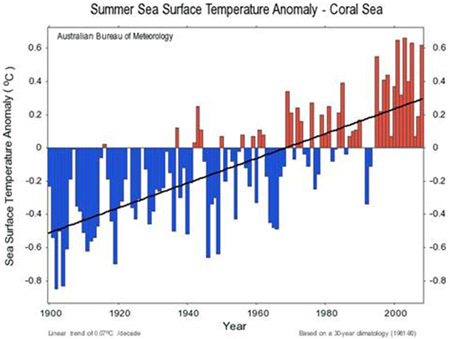
Figure 8. Changes in sea surface temperature over the past 100 years (Australian Bureau of Meteorology).
There has been some debate about whether or not reef-building corals are able to modify their sensitivity to temperature and thereby beat the climate change signal over the time. Evidence cited in favour of this hypothesis has been the observation that corals and some oceans are already experiencing very high sea temperatures. One example are corals growing in the Arabian Gulf, where sea temperatures reach 36°C. The problem with this example is that corals have adapted to their local temperature conditions, but over hundreds if not thousands of years. The ability of an Arabian Gulf coral to migrate and successfully establish a reef in the Indian Ocean over 10 or even 100 years is not feasible given what we know about the migration rate corals.
Another point of discussion has centred on the ability of corals to change their zooxanthellae for varieties that are more tolerant to temperature. This idea that led to the adaptive beaching hypothesis (Buddemeier; Fautin 1993) which proposed that corals bleached deliberately to swap one set of zooxanthellae for more tolerant zooxanthellae. So far, there has not been any compelling evidence that this hypothesis is true. Furthermore, as we have begun to further understand the intricate intracellular symbiosis that corals form with their dinoflagellate zooxanthellae, the more it has become clear that this symbiosis takes an enormous length of time to evolve. That is, to live inside another cell, you need to modify self recognition, integrate biochemically, and undergo a whole series of different changes to ensure that a harmonious endosymbiosis results. The idea that corals could form and use a basis in ecological timeframes of a few years is very unlikely given their long generation times.
Additionally, it is extremely unlikely that the thermal tolerance of reef-building corals comes down to that of its zooxanthellae. Heat stress is also likely to be a problem with the host coral, so changing its zooxanthellae is unlikely to solve the problem of rising sea temperatures (Hoegh-Guldberg 1999; Hoegh-Guldberg et al. 2002; Hoegh-Guldberg et al. 2007). This said, some corals do have multiple genetic varieties of zooxanthellae in their tissues. These can be shuffled such that the more thermally tolerant ones can dominate as sea temperatures rise. These separate symbiosis have all evolved with their coral hosts and consequently, provide only limited change in the thermal tolerance of the association as it is challenged by rising sea temperatures. That is, the changes are phenotypic not genetic, and hence do not allow corals to adapt optimally to the novel conditions that are presented by rising sea temperatures.
The conclusion after considering all of the science surrounding the response of coral reefs to changes in sea temperature suggest that coral-dominated reef systems are very likely to disappear from a tropical oceans if we do not reduce carbon dioxide emissions.
How does ocean acidification interact with warming sea temperature?
Coral reefs are susceptible to the impacts of ocean acidification (Raven et al. 2005). Ocean acidification arises from increased amounts of CO2 entering the world's oceans. CO2 reacts with water to create a dilute acid (carbonic acid), which interacts with other chemical species present in sea water such as carbonate ions. In the latter case, carbonate ions are increasingly turned into bicarbonate ions. The two changes brought about by ocean acidification, decreasing pH, and carbonate ion concentrations, impact a wide range of marine organisms (Hendriks et al. 2009; Kleypas and Langdon 2006; Kleypas et al. 2006). There is now a large amount of information showing that reef building corals are susceptible to relatively small changes in ocean acidification (Kleypas and Langdon 2006), probably as a result of declining carbonate ion concentrations in the first instance. An important point is that warming sea temperatures and steadily acidifying oceans interact in terms of their impact on reef building corals. That is, the thermal sensitivity of corals appears to increase as oceans become increasingly acidic (Anthony et al. 2008). The full implications of this interaction between climate change stressors has not been fully explored to date.
What evidence do we have that warming and acidification are affecting the Great Barrier Reef?
There is now very strong evidence that the Great Barrier Reef is being affected by the combination of these two stress factors. In a study of 328 long-lived corals from a full range of habitats on the Great Barrier Reef has revealed that calcification rates have fallen by 15% since 1990, which is unprecedented in the 400 years of coral core records examined (De'ath et al. 2009). Similar results have also been found in a number of sites around the world (Tanzil et al. 2009). While it is not possible to attribute this change to either increasing tropical sea temperatures or ocean acidification on their own, this study does reveal that changes to the conditions within the Coral Sea (which is steadily rising in sea temperature and acidity) are driving some exceptional changes to fundamental reef processes like calcification.
Is there a threshold above which coral-dominated ecosystems will disappear?
Other evidence of the importance of these two factors comes from our current understanding of what limits the growth and distribution of coral reefs worldwide. The current distribution of carbonate coral reefs around the world today is associated with concentrations of carbonate ions of 200 µmol per kilogram water or more. In this case, there is a natural gradient towards reduced carbonate ion concentrations and more acidic oceans at higher latitudes. This is primarily due to the fact that cold water can contain a lot more carbon dioxide and water.
The significance of this threshold for carbonate ions is that these are the concentrations that you get in tropical oceans when carbon dioxide increases above 450 ppm. Given that these levels of carbon dioxide in the atmosphere are likely to be associated with at least a 2°C increase in sea temperature, it appears that coral reefs will largely disappeared if atmospheric concentrations of carbon dioxide exceed 450 ppm. The full background to this argument can be found in the following review paper in Science magazine: (Hoegh-Guldberg et al. 2007). In terms of the distribution of waters that will have the right chemistry for carbonate coral reef ecosystems, the following set of diagrams from this paper outlines how these change as atmospheric levels of CO2 increase (number in top left hand side of each panel). The blue water in these diagrams is essentially that required for carbonate reef ecosystems (which are represented by the pink dots).
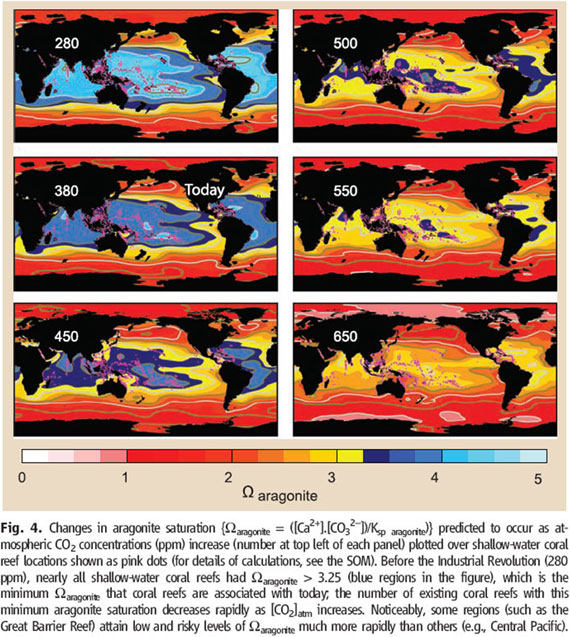
Figure 9. Changes in ocean chemistry is a function of atmospheric CO2 concentration. See Hoegh-Guldberg et al. (2007) for further details.
Corals survived in past climates with warmer temps and higher CO2 than now, so shouldn't GBR survive now?
The Great Barrier Reef has undergone major transformations in the geological past. During the glacial periods when large amounts of water have been locked up in glaciers and landlocked ice masses, sea levels were over 100 m lower than they are today. This meant that the Great Barrier Reef lagoon was completely exposed and was a wooded grassland not too dissimilar to those found in Queensland today. All of the islands and reef structures that remained after the water retreated were covered with soil and were unrecognisable as coral reefs.
This has prompted some to ask: Why are we so worried about climate change today if the Great Barrier Reef has undergone these types of changes in the past?
The answer to this question is straightforward and comes down to the issue of the rate of change. The changes in temperature and sea level associated with glacial cycles occurred over 10-20,000 years. In this period of time, global temperatures changed by 5-8 degrees Celsius and atmospheric concentrations of carbon dioxide by 100 ppm. If we compare these sets of changes to that going on today, we see that we are undergoing similar set of changes only we are undertaking them in less than 100 years. That essentially means that the rate of change today is about 100-200 times faster than in the past.
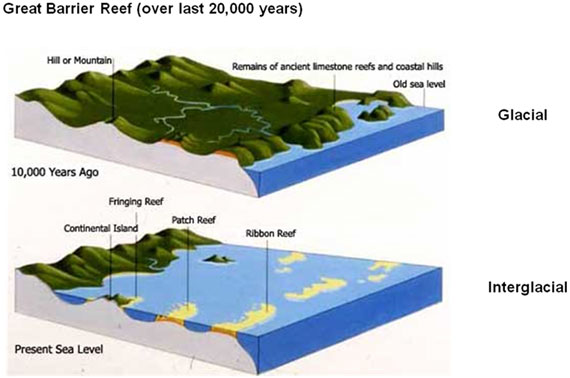
Figure 10. Changes in the Great Barrier Reef and Queensland coastline between glacial and interglacial periods. During last glacial period, sea levels were 100 m lower than they are today, exposing a large part of the Great Barrier Reef which subsequently covered by grasslands and forests.
While some changes in the past were relatively rapid, animals and plants like those associated with the Great Barrier Reef had time to shift their geographic distribution as the environments changed. Today, there are clear signs that the speed of change is exceeding the ability of organisms and ecosystems to shift. The Great Barrier Reef provides an instructive example of how fast reef ecosystems need to migrate if they were to keep pace with changing climate. Corals at the northern end are adapted to warmer temperatures (about 3°C) than those in the southern part of the Great Barrier Reef. If corals were to move southward at a rate which matched an increase in sea temperature of 3°C by 2100, they would necessarily have to travel the entire length of the Great Barrier Reef (2500 km) within approximately 100 years. That means that reef ecosystems would have to travel at the rate of 25 km per year. There is very little evidence that complex ecosystems such as the Great Barrier Reef would be able to travel at this impossibly high rate of movement of the environmental conditions necessary for Coral Reef development.
The final point that needs to be made with respect to the question of why we are worried today if coral reefs of undergone big changes in the past is that of timescale. When coral reefs have experience calamities in the past, they have been absent for very long periods (i.e. millions of years). These timescales are well beyond those that make any sense with respect to humans and their dependence on the ecological services provided by coral reefs.
Putting it bluntly, it is irrelevant that reefs have bounced back in geological time when the timescale of importance for the people depend on coral reefs (such as our vibrant tourist industry here in Australia, or the millions of people around the world need them for their food) is on a month to year basis.
References:
Anthony, K. R., D. I. Kline, G. Diaz-Pulido, S. Dove, and O. Hoegh-Guldberg, 2008: Ocean acidification causes bleaching and productivity loss in coral reef builders. Proc Natl Acad Sci U S A, 105, 17442-17446.
Bellwood, D. R., T. P. Hughes, C. Folke, and M. Nyström, 2004: Confronting the coral reef crisis. Nature, 429, 827-833.
Berkelmans, R., 2002: Time-integrated thermal bleaching thresholds of reefs and their variation on the Great Barrier Reef. Marine ecology progress series, 229, 73-82.
Berkelmans, R., and J. Oliver, 1999: Large-scale bleaching of corals on the Great Barrier Reef. Coral Reefs, 18, 55-60.
Berkelmans, R., G. De’ath, S. Kininmonth, and W. J. Skirving, 2004: A comparison of the 1998 and 2002 coral bleaching events on the Great Barrier Reef: spatial correlation, patterns, and predictions. Coral Reefs, 23, 74-83.
Bruno, J., C. Siddon, J. Witman, P. Colin, and M. Toscano, 2001: El Nino related coral bleaching in Palau, western Caroline Islands. Coral Reefs, 20, 127-136.
Bruno, J. F., and E. R. Selig, 2007: Regional decline of coral cover in the Indo-Pacific: timing, extent, and subregional comparisons. PLoS ONE. , 2, e711.
Buddemeier, R., and D. Fautin, 1993: Coral bleaching as an adaptive mechanism. Bioscience, 43, 320-326.
De'ath, G., J. M. Lough, and K. E. Fabricius, 2009: Declining Coral Calcification on the Great Barrier Reef. Science, 323, 116-119.
Done, T., P. Whetton, R. Jones, R. Berkelmans, J. Lough, W. Skirving, and S. Wooldridge, 2003: Global Climate Change and Coral Bleaching on the Great Barrier Reef. Final Report to the State of Queensland Greenhouse Taskforce through the Department of Natural Resources and Minings, QDNRM, Brisbane. http://www.nrm.qld.gov.au/science/pdf/barrier_reef_report_1.pdf and http://www.nrm.qld.gov.au/science/pdf/barrier_reef_report_2.pdf, Q. G. Department of Natural Resources, Ed.
Donner, S. D., W. J. Skirving, C. M. Little, M. Oppenheimer, and O. Hoegh-Guldberg, 2005: Global assessment of coral bleaching and required rates of adaptation under climate change. Glob Change Biol, 11, 2251-2265.
Dove, S. G., and O. Hoegh-Guldberg, 2006: The cell physiology of coral bleaching. Coral Reefs & Climate Change: Science and Management, 1-18.
Glynn, P. W., 1983: Extensive Bleaching and Death of Reef Corals on the Pacific Coast of Panama. Environmental Conservation, 10, 149-154.
Glynn, P. W., 1993: Coral reef bleaching: ecological perspectives. Coral Reefs, 12, 1-17.
Glynn, P. W., 1996: Coral reef bleaching: facts, hypotheses and implications.
Glynn, P. W., and L. Dcroz, 1990: Experimental-Evidence for High-Temperature Stress as the Cause of El-Nino-Coincident Coral Mortality. Coral Reefs, 8, 181-191.
Hendriks, I., C. Duarte, and M. Álvarez, 2009: Vulnerability of marine biodiversity to ocean acidification: A meta-analysis. Estuarine, Coastal and Shelf Science.
Hoegh-Guldberg, O., 1999: Climate change, coral bleaching and the future of the world's coral reefs. Mar Freshw Res, 50, 839-866.
Hoegh-Guldberg, O., and G. J. Smith, 1989: The effect of sudden changes in temperature, light and salinity on the population density and export of zooxanthellae from the reef corals Stylophora pistillata and Seriatopra hystrix. Journal of Experimental Marine Biology and Ecology, 129, 279-303.
Hoegh-Guldberg, O., R. J. Jones, S. Ward, and W. K. Loh, 2002: Communication arising. Is coral bleaching really adaptive? Nature, 415, 601-602.
Hoegh-Guldberg, O., and Coauthors, 2007: Coral reefs under rapid climate change and ocean acidification. Science, 318, 1737-1742.
Jones, R., O. Hoegh-Guldberg, A. Larkum, and U. Schreiber, 1998: Temperature induced bleaching of corals begins with impairment to the carbon dioxide fixation mechanism of zooxanthellae. Plant, Cell and Environment, 21, 1219-1230.
Jones, R. J., and O. Hoegh-Guldberg, 1999: Effects of cyanide on coral photosynthesis: implications for identifying the cause of coral bleaching and for assessing the environmental impacts of cyanide fishing. Marine Ecology Progress Series, 177, 83-91.
Kerswell, a., and R. Jones, 2003: Effects of hypo-osmosis on the coral Stylophora pistillata: nature and cause of'low-salinity. Marine Ecology Progress Series, 2003 ;0(0):0.
Kleypas, J. A., and C. Langdon, 2006: Coral reefs and changing seawater chemistry, Chapter 5 Coral Reefs and Climate Change: Science and Management. AGU Monograph Series, Coastal and Estuarine Studies, J. Phinney, O. Hoegh-Guldberg, J. Kleypas, W. Skirving, and A. E. Strong, Eds., Geophys. Union, 73-110.
Kleypas, J. A., R. A. Feely, V. J. Fabry, C. Langdon, C. L. Sabine, and R. R. Robbins, 2006: Impacts of ocean acidification on coral reefs and other marine calcifiers: A guide for future research, Report of a workshop held 18-20 April 2005, St Petersburg, FL, sponsored by NSF, NOAA and the US Geological Survey, 88pp.
Koop, K., and Coauthors, 2001: ENCORE: The Effect of Nutrient Enrichment on Coral Reefs. Synthesis of Results and Conclusions. Marine Pollution Bulletin, 42, 91-120.
McCulloch, M., S. Fallon, T. Wyndham, E. Hendy, J. Lough, and D. Barnes, 2003: Coral record of increased sediment flux to the inner Great Barrier Reef since European settlement. Nature, 421, 727-730.
Oliver, J. K., R. Berkelmans, and C. M. Eakin, 2009: Coral Bleaching in Space and Time. Coral Bleaching, 21-39.
Pandolfi, J. M., and Coauthors, 2003: Global trajectories of the long-term decline of coral reef ecosystems. Science, 301, 955.
Raven, J., and Coauthors, 2005: Ocean acidification due to increasing atmospheric carbon dioxide.
Reaka-Kudla, M. L., 1997: Global biodiversity of coral reefs: a comparison with rainforests. Biodiversity II: Understanding and Protecting Our Biological Resources, M. L. Reaka-Kudla, and D. E. Wilson, Eds., Joseph Henry Press, 551.
Sweatman, H., S. Delean, and C. Syms, 2011: Assessing loss of coral cover on Australia’s Great Barrier Reef over two decades, with implications for longer-term trends. Coral Reefs, 2, 521-531.
Tanzil, J., B. Brown, A. Tudhope, and R. Dunne, 2009: Decline in skeletal growth of the coral Porites lutea from the Andaman Sea, South Thailand between 1984 and 2005. Coral Reefs, DOI 10.1007/s00338-008-0457-5.
Wachenfeld, D., 1997: Long-term trends in the status of coral reef-flat benthos-The use of historical photographs. 134-148.
Advanced rebuttal written by Ove Hoegh-Guldberg
Update July 2015:
Here is a related lecture-video from Denial101x - Making Sense of Climate Science Denial
Additional videos from the MOOC
Expert interview with Annamieke Van De Heuvel
Expert interview with Gregg Webb
Last updated on 6 July 2015 by pattimer. View Archives































 Arguments
Arguments




























The Queensland Government has just approved the Carmichael Coal Mine. As James Hansen has long pointed out, continuing mining of coal will push as well beyond the 2 C target. On a related note, the Great Barrier Reef this year is suffering just its eigth ever mass coral bleaching, the first having been in 1981. This is part of just the third global mass coral bleaching, the first having been in 1998.
This is unquestionably the result of global warming. The three global events have all occurred in years when Global Mean Surface Temperature has approximated 1 C above preindustrial levels, yet as a best case we will allow global temperatures to rise by another 0.5 C above that level, and the international target is another 1 C above that level. To keep temperatures below 2 C we need to phase out the use of coal. Phasing out the use of coal means approving no more coal fired power plants, and above all, no more coal mines.
That means the Queensland Government have just, in effect, approved the destruction of the Great Barrier Reef. I ask that you, as I did earlier, contact the Queensland Premier to register your disgust at that short sighted decission.
In the article, it mentioned that CO2 dissolves in water and causes ocean acidification. Therefore, the CO2 amount are highly found in both atmosphere and ocean? If so, then GBR is really in big trouble because this is a big synergy caused from both the global warming and ocean acidification. Am I understand correctly?
Ping34 @ post #2 , you are correct. The Great Barrier Reef has suffered much damage from (mostly) the increasing water temperatures. And it will get worse, because global warming is continuing, and the politicians of the world are making only minor effort to counter the effects of CO2 emission.
The GBR is large — about the size of Italy — and not all of its beauty has been destroyed (so far). The local tourist industry does not like to mention the damage, or that the damage will almost certainly increase over the next decades. And Australians do not like to think about the situation. Also, other coral reefs in other places, are being damaged. But the GBR is the most important and beautiful of them. Ping34, if you wish to see the GBR, you should visit it as soon as you can. Do not wait for 10 or 20 years.
There is one hope. Scientists are studying ways of selective breeding of corals which are more resistant to high temperatures. It is uncertain how much success they will have; or how well they can re-populate such a large reef in just a few decades; or if they can re-create the numerous varieties of corals which give the reef its beauty and provide the habitat for the many beautiful varieties of fish.
https://www.youtube.com/watch?v=m63KMzL1jzs
[BL] It is inappropriate to add comments consisting only of a link. Please read the Comments policy.
No link or picture only. Any link or picture should be accompanied by text summarizing both the content of the link or picture, and showing how it is relevant to the topic of discussion. Failure to do both of these things will result in the comment being considered off topic.
https://www.youtube.com/watch?v=yHF6MJZTCY8
[BL] It is inappropriate to add comments consisting only of a link. Please read the Comments policy.
No link or picture only. Any link or picture should be accompanied by text summarizing both the content of the link or picture, and showing how it is relevant to the topic of discussion. Failure to do both of these things will result in the comment being considered off topic.