SkS Analogy 13 - Water glasses and Greenhouse gases
Posted on 17 July 2018 by Evan, jg
Tag Line
How do you add water to a full glass?
Elevator Statement
- Adding water to a full glass causes the water to rain over the sides.
- To add water to a full glass, first increase the height of the glass.
- Then add more water to the larger glass.
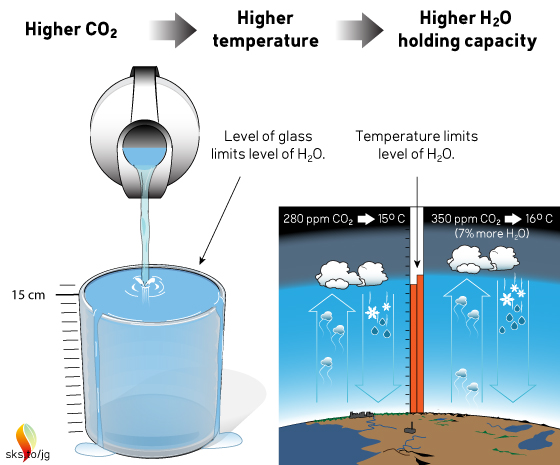
In the atmosphere this process looks as follows.
- Adding water vapor to saturated air causes mist, clouds, or rain to form.
- Adding greenhouse gases to the atmosphere causes warming, raising the dew point.
- The higher dew point allows more water vapor to be added to the air.
Climate Science
Water is an important greenhouse gas that is stronger than CO2 in its per/molecule warming effect. Even though water is a stronger greenhouse gas than CO2, CO2 controls the amount of water vapor in the air. CO2 is the main control knob, because at a constant average global temperature, it is impossible to add extra water to the atmosphere. If you do, it simply falls out as rain. Unlike water, there is no limit to how much CO2 we can add to the Earth’s atmosphere.1 Adding CO2 causes temperature to increase, and at the increased temperature air holds more water vapor.2 See Appendix A for additional discussion of this relationship.
There are many similar relationships
- The flick of a small switch controls large electrical current.
- The volume control on a stereo system controls the booming sound levels.
- A car’s accelerator pedal controls hundreds of horsepower.
- Throttles on an airplane control 10’s of thousands of pounds of thrust.
- CO2 levels in the atmosphere control H2O levels by first increasing temperature, and the increased temperature increases the dew point.3
Like a glass of fixed volume, as long as the global average temperature is unchanging, the water vapor held in the atmosphere is unchanging.
Increasing the height of a 15-cm tall glass by 1 cm allows the glass to hold 7% more water.
Increasing the average temperature of the Earth from 15°C (pre-industrial temperature) to 16°C (average temperature as of 2017) allows the atmosphere to hold 7% more water. More water vapor means heavier rainfall and stronger storms.
Appendix A
There are two points to clarify in this analogy.
- The concentration of water vapor in the atmosphere is controlled by atmospheric temperature.4
- Currently the long-term increase of average atmospheric temperature is primarily controlled by CO2.5
The concentration of water vapor in the atmosphere is controlled by atmospheric temperature. It does not matter how the temperature increases, whether due to greenhouse-gas (GHG) emissions, whether due to stronger solar insolation, whether due to urban heat-island effects on steroids, or whether due to some other effect. Excessive vulcanism can lead to cooling, which, because of the decreased global temperatures, will cause a decrease of water-vapor concentration. If global average temperatures increase, for whatever reason, this increases the amount of water vapor the air can hold: about 7% more water vapor for each 1°C of warming. This is the simple, physical relationship between average global temperature and water-vapor content described by the Clausius-Clapeyron equation.
For warming due to GHGs, there is a different relationship between CO2 concentrations, H2O concentrations, and the eventual warming. When CO2 is initially added to the atmosphere, there is a small amount of warming due to the increased CO2 concentrations. This warming raises the amount of water vapor the atmosphere can hold (due to the Clausius-Clapeyron equation), which increases the warming further. This feedback looks like …
- Increased CO2 concentration increases warming (greenhouse effect)
- Increased warming increases water vapor (Clausius-Clapeyron equation)
- Increased water vapor increases warming (greenhouse effect)
- Increased warming increases heat loss from atmosphere due to radiation
- Temperature increases until processes 1 to 4 are balanced
Whereas there is a simple relationship between increased global average temperature and atmospheric water-vapor content, the feedback between CO2, H2O, global-average temperature, and radiation heat loss is more complicated. This Water-glass analogy states that CO2 is the controller of atmospheric water-vapor concentration, because the main driver of warming currently is CO2 emissions. If we lived in a period of strong solar increase or decrease, then the analogy might be modified to show the sun as the main controller of atmospheric water-vapor concentration. Or if we lived in a period of high vulcanism, then we might indicate that SO2 emissions were the main controller of atmospheric water-vapor content, because SO2 in the atmosphere leads to cooling.
So the point is that whereas the relationship between temperature and average water-vapor content is a fundamental, physical relationship governed by Clausius-Clapeyron equation, the role of CO2 as the main controller of water-vapor content is due to the fact that the main driver of global warming is currently CO2 emissions.
Footnotes
1. Technically there is a limit, which is the point at which the atmosphere is pure CO2. But within any limits that still sustain life on Earth, there is no limit to how much CO2 we can add to the atmosphere.
2. According to the Clausius-Clapeyron equation, for every 1°C average warming, the atmosphere holds about 7% more water vapor. For a good discussion of the relationship between CO2 and H2O, see this Forbes article.
3. Relative humidity stays about the same, but at a given relative humidity and higher temperature, the water vapor in the air increases, according to the Clausius-Clapeyron equation.
4. The reference here is specifically to the relationship between average water concentration in the atmosphere related to average global temperature. In areas like deserts, the temperature can be very high, but the atmospheric water-vapor concentration very low. The temperature limits the maximum water-vapor holding capacity of air, and given a relatively fixed ratio of arid and non-arid places in the world, the average water-vapor concentration of the atmosphere remains strongly correlated to average global temperatures. This is particularly true because even though deserts present an exceptional landscape where humidity is low, about 75% of the surface of the Earth is covered by water.
5. We specifically refer to long-term average temperature increases, because year-to-year variability is still controlled by other variables in the climate system such as El Nino/La Nina cycles, solar cycles, air pollution levels, volcanoes, etc. But the long-term increase has been linked to rising CO2 concentrations with a high degree of confidence (see Ocean Time Lag).































 Arguments
Arguments






























I found it confusing for me reading an analogy followed by the physical laws when normally its around the other way around. But the analogy is good and the article is very clear.
I would suggest a link to the CC equation for us non physicists to examine or to refresh memories. Although its easily googled, so It's not a big issue.
Another related point is CO2 stays in the atmosphere for ages while water vapour has a short residence time, another reason why CO2 is regarded as the control knob.
nigelj@1 Thanks for your suggestions. We will work them into a future version. I added a link for the CC equation. I did not use the Wiki link, because I think it is too technical for non-specialists
Every morning, my lawn is covered with H2O from the atmosphere.