From the eMail Bag: CO2 in the air and oceans
Posted on 6 December 2016 by David Kirtley
We occasionally receive excellent questions and/or comments by email or via our contact form and have then usually corresponded with the emailer directly. But, some of the questions and answers deserve a broader audience, so we decided to highlight some of them in a new series of blog posts. This is the first one of those posts and more will follow every once in a while in the future.
We recently received an email from Jeffrey Middlebrook who asked about the dynamics of CO2 transport from the atmosphere to the oceans:
...as atmospheric water vapor increases with CO2-driven atmospheric warming, there will be more CO2 capture by the increased water vapor (yielding more carbonic acid) which will transfer more CO2 to the oceans, thereby decreasing the effects of atmospheric CO2 as a greenhouse gas, and with greater precipitation due to more atmospheric water vapor more atmospheric heat will be transferred to the oceans and terrestrial landscapes. It seems plausible that the increases in atmospheric water vapor due to increases in atmospheric CO2 might just produce a strong negative feedback.
About 46% of human emissions of CO2 stay in the atmosphere, while ~26% makes its way to the oceans, and ~28% is used by plants. Our emissions of CO2 may be good for plants (at least for now) but the additional CO2 in the oceans is leading to climate change's "evil twin": ocean acidification. (For much more about ocean acidification see our series: OA is not OK.)
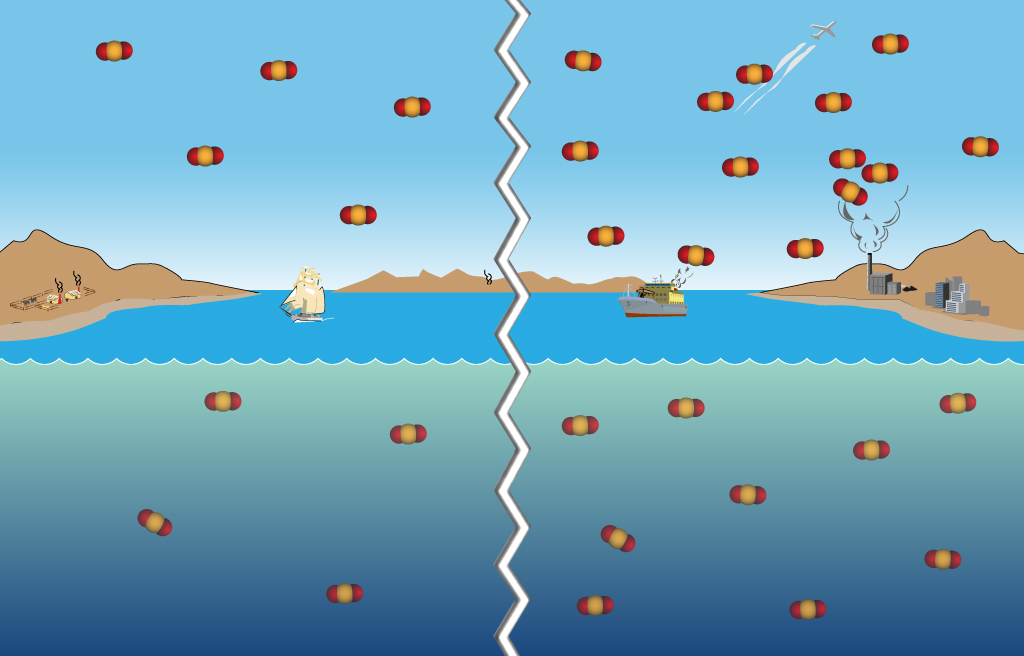 Figure 1. CO2 added to the atmosphere is absorbed by the oceans due to Henry's law. More CO2 added since pre-industrial times leads to more CO2 absorbed by the oceans.
Figure 1. CO2 added to the atmosphere is absorbed by the oceans due to Henry's law. More CO2 added since pre-industrial times leads to more CO2 absorbed by the oceans.
Think of how large the world oceans are, and the vast ocean surface in contact with the equally vast atmosphere. The oceans primarily absorb CO2 directly from the atmosphere because of the differences in partial pressure across the ocean's "cool skin" layer. If there is more CO2 in a parcel of air than there is in a parcel of water, then the water absorbs more CO2 until there is an equilibrium between the two (Henry's law, see Figure 1). Temperature also plays a role since warmer water holds less CO2 than cooler water.
Another consequence of climate change is the increased capacity of warmer air to "hold" more water vapor. Jeffrey's comment suggests that as more water vapor condenses and forms precipitation more CO2 will be absorbed in precipitation and eventually enter the oceans by this route. Henry's law still operates between the atmosphere and suspended precipitation so there is a bit of CO2 absorption. But the volume of precipitation, say in a massive thunderstorm, is much smaller than the volume of the ocean surface, so this is a minimal effect. The vast majority of CO2 in the oceans gets there directly from the atmosphere via Henry's law.
Absorption of CO2 by the ocean is constrained by the slow, over-turning circulation of ocean waters.
With the oceans absorbing nearly a quarter of our CO2 emissions, will they work to lessen climate change's impacts in the future? Jeffrey alludes to this in his sentence, "It seems plausible..."— more water vapor "scrubbing" more CO2 out of the atmosphere and into the oceans, leaving less and less of an enhanced greenhouse effect in the atmosphere, leading to less and less warming. Unfortunately this scenario takes a very long time--about a thousand years--to play out (see video). Remember that the transport of CO2 from the atmosphere takes place at the ocean's surface which saturates the upper layer of the ocean with CO2. The deeper layers of the ocean are essentially cut off from this exchange. Slow-moving ocean circulation eventually brings fresh, deep water to the surface where it can absorb CO2 from the atmosphere, but this process is too slow to keep up with our emissions.
All the more reason to decrease our emissions of CO2 by switching from fossil fuels to more sustainable energy systems.
Thanks to Daniel Bailey, Gunnar Schade and Glenn Tamblyn for help in answering Jeffrey's email. And to jg for his excellent graphics and video.































 Arguments
Arguments






























How does the Ocean fight acidification?
As far as I'm aware one of the few remaining uncertainties about climate change is the exact nature of some of the positive and negative feedbacks. It does appear on the best evidence available that positive feedbacks outweigh negative feedbacks. Given periods of warming in the past last a long time this is evidence positive feedbacks dominate over negative ones.
It appears that positive feedbacks may be dominant in the short to medium term (we are already seeing positive feedbacks from the Arctic) but periods of warming do not seem to be permanent. Perhaps negative feedbacks like elements of the carbon cycle may kick in much longer term (over millenia). This is too long term to benefit humanity.
@ nigelj
Debatable. Obviously humans can effect the results. Rather than accidently triggering the reinforcing feedbacks (which is what we have done so far), there is always the option of purposely triggering the stabilizing feedbacks.
Red Baron @3, that is a good thought but do you have any examples?
RedBaron, nigelj,
THere are no examples of such feedbacks. As you correctly asserted @2, the stabilising feedbacks act on a long timeframe of millenia. Ultimate known negative feedback of rock weathering acts on 100ky+ timeframe. Such timeframe is by definition irrelevant to humans and to human civilisations. Talking about "reinforcing" those feedbacks, while cranking the main control knob (CO2) to the overheat rate never seen in many milion years is just like joking. A sweet fairy tale, but the physics & the numbers tell you it's not a real tale.
There are case studies, mostly from Australia and CSIRO and a few from USDA that indicate between 5–20 t COe /ha/year can be sequestered long term in the soil via the newly discovered “liquid carbon pathway”. Basically to simplify a very complex and exciting new line of research, the LCP goes like this: Photosynthesis to root exudates to mycorrhizal fungi to glomalin to humic polymers to mollic epipedon. This can apply to restoring both degraded grasslands/savannas and/or seasonally arid cropland, under the right conditions, management, and methods of course. If you do that math, conservatively one could sequester long term into the soil between 62% and 250% of current yearly emissions.
So the answer is potentially possible with a relatively high uncertainty. Since 250% is more than emissions, this stabilizing feedback could draw down CO2. That means yes it could work and we now know how to trigger it. Certainly the soil sink is large enough to hold all that carbon long enough to give the oceans a break and chance to catch up. Since it is a biological feedback that evolved over millions of years, it is unlikely to have an unacceptable risk level. We are after all simply restoring the lost ecosystem services on almost 1/2 the planet land surface that WE naively degraded with agriculture. The biggest risk being we might do it too well and slip back into a glaciation phase. But I suspect we would be quite capable of preventing that.
However, there are some nuances. Likely if we draw down CO2 too rapidly the oceans might become an emissions source too. Like a fizzing coke! That means while we probably can sequester that carbon from the atmosphere, it might not actually do as much to stop AGW as anticipated due to CO2 being replaced almost as fast as we draw it out. Another nuance is that the low end is 62%. That wouldn’t be enough. That means we must also drop emissions to at minimum 62% or less of what they are now by converting as much as possible to renewables like solar and wind etc…
Methane is not a problem. This will actually help the methane problem significantly due to restored methanotrophic activity. Yes, it’s true. Raised properly and as part of a healthy grassland biome, cows actually help lower atmospheric methane. The problem with methane isn’t the cow. Not their fault. It’s the feedlots and the vast overproduction of corn and soy used to supply those feedlots, thawing permafrost, dead zones in the ocean, fossil fuel well leakage etc….
Soil Microorganisms as Controllers of Atmospheric Trace Gases (H2, CO, CH4, OCS, N2O, and NO)
Grazing management impacts on vegetation, soil biota and soil chemical, physical and hydrological properties in tall grass prairie
Restoring the climate through capture and storage of soil carbon through holistic planned grazing-White Paper
Doesn't the evaporation - condensation - rain cycle transfer heat from the ocean to the atmosphere?
In his email, Middlebrook claims the opposite. "...with greater precipitation due to more atmospheric water vapor more atmospheric heat will be transferred to the oceans and terrestrial landscapes."
Well done, RedBaron, for bringing up the restoration of natural levels of carbon in the soil as a powerful mitigation measure. Awareness of this seems to be growing. So far, I have not seen enough peer reviewed evidence that we could replace coventional agriculture and still get the same productivity while increasing soil carbon percentages long term but there is plenty of exciting relatively woo-free anecdotal evidence from such as Drs Christine Jones, Elaine Ingham and David Johnson.
Nick,
If you use vertical stacking and polycultures you can almost guarantee increased yields per acre. Productivity is a different issue, that is yields per man hour. No we generally have not succeeded yet in beating the industrial production models there. I would point out though....is that the problem it once was? Don't we have plenty of people capable and willing to work on this planet? It is not the old days where labor shortages were the main priority due to massive wars and famines and we were forced to import cheap labor just to keep up. (slaves from Africa, and Chinese laborers for the railroads...etc.... as examples from my country)
The HUGE fish-hook with industrial production models is that while they may have spectacular and essential yields now, they are rapidly destroying the globe's soils. The FAO were predicting only 60 more harvests before they have been wiped out and even that figure's impact is illusionary...it won't be a case of 60 years and 'click' the light goes out, it will be decades of declining output and increasing starvation with all the social disruption that will (and already is) entail.
sailingfree asked
Doesn't the evaporation - condensation - rain cycle transfer heat from the ocean to the atmosphere?
Exactly right, sf, sensible heat at the surface is converted into latent heat through evaporation. That heat is then transported upward by convection and released as sensible heat higher up in the atmosphere by condensation, so that part of Jeffrey Middlebrook's question is based on an incorrect understanding of atmospheric heat transport.
Red Baron @6, soil absorption of carbon intuitively sounds plausible to me and clearly you have some genuine expertise. However I'm sure I read an article recently that a warming climate actually causes increased microbial activity that actually results in soils releasing CO2? You would need to prove which factor dominates.
Your confusion results from not clearly defining the carbon in the soils. Actually both are correct even though they may seem contradictory. The simple answer is this. There are two completely different biochemical pathways. The first we will call "labile carbon". This is the carbon resulting from the decay of organic plant and animal debris in the top O-horizon and the upper layers of the A-horizon of the soil. Bacteria, fungi, small arthropods, worms etc... do decay that material more rapidly as temperatures warm. This carbon mostly ends back into atmosphere as CO2, or CH4 in anaerobic conditions. The Roth C mathematical model is pretty effective at determining the tiny % that gets stored long term into the soil this way. It isn't much. This whole process therefore is considered a reinforcing feedback, rather than a stabilizing feedback.
The newly discovered LCP feeds an entirely different soil carbon pool which we can call the "stable carbon" fraction of the soil carbon pool. Instead of a near zero net, this carbon has an overwhelming tendency, under the right conditions, to even increase as temp or CO2 increases. Here is why. The C4 photosynthesis pathway is primarily your warm season grasses, rather than cool season grasses or trees. It is far more efficient, all else equal. So more root exudates to feed AMF. Glomalin has properties similar to "super glue" and while it does decay slowly, that decay is deep in the soil profile rather than on the surface. A far greater % converts to humic polymers that tightly bind to the mineral substrate. If anything, as it gets hotter and drier, the roots go deeper seeking ground water and even a greater % of that carbon ends up stable. Same with CO2 fertilization. For these types of plants, CO2 increases the carbon that follows this pathway. So you start with more and sequester a higher % of that more deep in the soil long term. This is a very different outcome from the decay of debris on the surface. One is a stabilizing feedback, the other a reinforcing feedback.
Remember, my premise was we should purposely trigger a stabilizing feedback, rather than the triggering of the reinforcing feedbacks we are accidentally triggering now. So obviously I do understand the reinforcing feedbacks are present now. It’s the main reason the “CO2 fertilization” effect isn’t really doing much for us. Whatever we gain is immediately offset by increased decay rate. But change the pathway to the LCP and CO2 fertilization actually helps to increase the rate of long term sequestration.
Eager Jim@11, me @7
Thanks. So no negative feedback afterall.
me@11
Not negative per the Middlebrook email. I am not referring to Red Barron's very interesting possibility.
Red Baron @13, thanks. I did think both processes were operating in tandem, hence my comment on which one you thought would be dominant. Looks like its carbon storage. Maybe you should write a research paper or article on it, as you are passionate about it.
Seems like soil carbon storage is proof at least a few negative feedbacks could be reinforced, but the problem is it would be a long term project and would require changes in farming techniques and I dont see that happening too fast. It seems hard enough persuading the world to reduce emissions. However I suppose every effort helps.
Another negative feedback is the forces that ultimately generate ice age cycles by reducing solar irradiation patterns on the earth. However the only way of reinforcing that would be geoengineering, and it would be very high risk.
We come back to carbon emissions as the main thing that can be realistically tackled in a practical sense. But the "politics" of the whole thing are the hard part.
nigelj @16
I seem to remember you live in New Zealand. So have a look at the Listener magazine for 19-25 November — it has an article on pasture cropping that is relevant.
RedBaron @13
I haven't forgotten my study of the links you gave me earlier to do with the aforementioned topic — an unexpected sojourn in hospital got in the way, but I hope to resume soon.
RedBaron
The first problem with the soil carbon theory is that It requires an intensity of effort and a quantity of compostible biomass far greater than that available in our dry land.
The second problem is that it is sequestering carbon, as also does the ocean cycle, in a way that makes this carbon permanently unavailable for human civilization to use in the future hundreds and thousands of years.
We are so hooked on the notion that there is abundant carbon and forever, not true. The Earth has been bleeding carbon off into space for billions of years, and it was only by chance that algal life set to work capturing carbon from the primative atmosphere and sequestering it in the form of coal, oil gas, and clathrates, forms of concentrated carbon that can be collected fairly easily by civilization. All along the way atmospheric CO2 has been reducing due to the bleed off and dispersion throughout the Earth's surface to the point where it is a trace gas at a level that still sustains life. Atmospheric CO2 in the natural cycle is topped up from the plate techtonic volcanic cycle as subducting plates boil off CO2 as rock moves into the mantle so we are not going to run out of atmospheric CO2 any time soon, but what we are at risk of running out of is readily available condensed Carbon, Carbon which our civilization would not function without, and I am not referring to carbon as fuel here.
So while you are wishing away our Carbon into the soil and oceans so that inefficient fossil fuel entrepreneurs can be rich for a century more be aware that at the end of the road there is neither enough carbon to build our technology, nor the carbon as fuel to process it.
I am 100% certain that the notion that "soil sequestration can absorb Australia's CO2 emissions" will not stand up to quantified scrutiny. It is a total false assumption that farmers who operate on a commercial razors edge will somehow have the extra resource to collect all of their biomass, compost it, and disperse it over any significant percentage of Australian lands under appropriate conditions in order to promote the growth of different soil fungi with subsequent CO2 sequestration. Most of Australia's land does not produce enough biomass to even begin to activate such a plan. The only thing I agree with you on, is that methane form cows is a bloated argument in my opinion.
The only poath forward for our civilisation is to agressively develop and implement sustainable and renewable technologies in order to reduce the rate of extraction of fossil carbon to both reduce atmospheric CO2, and preserve as much readily available carbon for the continuity of our civilisation.
Every single Australian is currently consuming a continuous 7 kilowatts of energy 24/7/365. That is a staggering energy consumption and all of that comes from fossil fuels. That is the equivalent of burning .75 litres of petrol every hour continuously. And that does not include the energy content of the goods Australia imports.
BilB,
Arbuscular mycorrhizal fungi (AMF) do not decompose compost. You are confused between AMF and Saprophytic fungi. So pretty much everything you wrote applies to something completely different. Furthermore, every example I gave in this and many other posts all went to this form of agriculture not to save the planet by sequestering carbon, but rather because they were backed into a corner and needed to find a more profitable way to farm or lose the farm. Every one. So that part of your analysis is completely wrong too. In fact the reason it is catching on and spreading so rapidly is that soil degradation is a more emmediate threat for many farmers than AGW. Turns out that there is more carbon missing from our soils than extra in the atmosphere. But your analysis is completely backwards because you don't understand the soil or the carbon cycle in the soil.
Well, Red Baron, from my read of Christine Jones' article the humification process is fostered with, amoung other things, composted material. I don't doubt the science at all, some of this science is covered in a very interesting read "the hidden Life of Trees", what I do doubt is the scale and reliability of the process en masse. The soil works the way it works because that is what the conditions allow. Christine Jones makes the sweeping claim that 60% of Australia's land area is used for food production, but that is a flawed claim as most of that land area is sparcely utilised by livestock and in no way ever travelled over by humans let alone intensively farmed in the way to promote the soil changes proposed.
The other problem I have with this approach is that you only get to store carbon in this intensive farming way after you've cleared away the 100 to 640 carbon tonnes of bush land vegetation that was there in the first place, so improving the soil carbon is really a restorative process rather than the creation of new carbon storage capacity. Yes it is a good thing to do, but it is really two steps back for one step forward, and having decided to clear land for farming improving soil carbon sequestration in this way should be a high priority, for all of the reasons you have mentioned.
RB, Excuse the density. In Lal, 2008 I see: "Strategies to increase the soil carbon pool include soil restoration and woodland regeneration, no-till farming, cover crops, nutrient management, manuring and sludge application, improved grazing, water conservation and harvesting, efficient irrigation, agroforestry practices, and growing energy crops on spare lands."
As you point out, all of these practices matter for the sake of the soil, but in terms of C sequestration WRT climate, they all amount to LULCC practices. So what is new? There doesn't seem to be anything controversial in any of the ideas proposed, but the mechanisms require actions which are already seen as being desirable, so where does this lead us?
BilB,
About 22.3 million hectares are planted annually to commercial grain crops across Australia.[1] At the rate Ms Jones measured in her pasture cropping case studies of 5-20 tCO2/ha/yr that equates to between 111.5 to 446 Mt CO2/yr. Australia emitted 549.3 Mt CO2 in 2014. So just pasture cropping alone could offset between 20% and 80% of Australia's CO2 emissions before we even begin to discuss other agricultural areas. That land was already cleared long ago, and the soils already degraded long ago.
As for the rest of your post regarding livestock grazing alone, a far larger land area, what are you refering to? Livestock? or properly managed livestock?
@Fergus Brown,
You asked "where does this lead us?". I have made a rough white paper on another thread here: That's just my idea. I highly suspect there are others who could flesh it out better, or suggest other ways forward.
PS to the post above. I found a way to allow Dr. Jones explain it herself, much better than I have communicated it, in an interview.
Dr. Christine Jones Explains the Life-Giving
Link Between Carbon and Healthy Topsoil
According to the 2015 Global Carbon Budget Report, cumulative land use emissions since 1750 from deforestation and soil deterioration amount to about 200 GtC. Presumably, the Holocene biosphere carbon stock was relatively stable prior to that (I know, Ruddiman argues for anthropogenic emissions before that, but they are not comparatively very big). If we are to stabilize the climate and don't bring fossil fuel emissions down to zero, we will need to sequester more than that.
My question, and it's a real one, I don't know the answer: is it reasonable to assume that we could store more carbon in the biosphere—in a stable manner and securely for the long-term—than there was there prior to human interference?
That is a great interview, Red Baron. So now what I see is yet another Australian great agricultural intellect attempting to educate farmers on how to improve their production with good knowledge and good practice.
Who knows, maybe Christine Jones will be the one who breaks through that tough farmer hide and actually get farmers changing their methods and understandings. The experience seems to be the same as talking global warming to tea party republicans. There has been an impressive procession of others ahead of her.
So as Fergus Brown says, get to it,...make it happen. It should, after all be a doddle to absorb all of Australia's emissions in the land. We do, afterall, have 30 Australian hectares per person, how hard can it be? Please let us know when it is done.
Andy Skuce,
You said,
We probably could. Maybe not. However, there probably is no need. This is the most common mistake made when I talk to climate scientists rather than farmers. Because your main focus is on the atmosphere you automatically equate loss of soil carbon as emissions. That is a portion of the loss yes. However, a far greater loss of carbon has resulted from erosion. That carbon did not all result in increases of atmospheric CO2, but rather most is currently sitting on the bottom of our waterways, lakes and oceans.
So we have the counter-intuitive result that actually long before we restore our agricultural soils to historic levels prior to human interference, that excess CO2 in the atmosphere will be long gone.
That leaves us back to the nuance I mentioned before. The Oceans. They will likely become an emissions source under any atmospheric draw down scenario. But I have seen no studies that looked closely enough at that to actually quantify it, neither by rate, nor by total. There are a few that have done work on how much they absorbed, but not what they might cough back up. Those numbers shouldn't be assumed to be equal, as many processes sequester carbon long term in the oceans, and fossil releases like methane clathrates can happen too. So quite honestly I don't know and it boggles my mind to even make an educated guess.
I suspect that the excess atmospheric carbon combined with additional atmospheric carbon released from the ocean will not be enough to restore the carbon to historical levels in the soil, but it probably should be enough to at least get our soils healthy and stable again. However, that is not an educated guess, not even a wild guess, just a hunch and a promising new line of research for someone smarter and younger than me. I have spent years just trying to understand the atmosphere and soil; The oceans and seabed? Way out of my league. The little I do know from talking to marine biologists only made me realise I basically know nothing. It is that complex.
Red Baron,
James Hansen has discussed ocean outgassing of CO2 once emissions become low enough that the ocean has more CO2 than the air. Look for articles on geoengineering that remove CO2 from the air. As I recall (sorry no link but you rarely link), the top 1000 meters of the ocean starts to emit CO2 quickly after CO2 in the atmosphere starts to drop and will be a very large amount of carbon dioxide compared to the amounts you are talking about sequestering in the soil. The ocean contains much more carbon than the atmosphere.
You need to provide data and/or links to support your claims that soil can sequester the amount of CO2 that will come out of the ocean. The immense amount of carbon that needs to be sequestered is the problem for any geoengineering scheme that proposes to remove carbon from the air. Most of the carbon that has been emitted would eventually have to be removed, natural sequestration processes are very slow on a human timescale. You do not appear to have determined the amount of carbon that your scheme needs to address.
At the same time the deep ocean absorbs some CO2. Depending on the exact amounts of emissions, sequestering and time frame the distribution of CO2 is different. Evaluating the issue is complex. One issue with soil sequestration is that if there was a change in soil use, say there was a severe drought or war, that can release the carbon sequestered in the soil.
Any sequestering of CO2 is a good thing and increased carbon in the soil increases productivity. I see little downside or risk to increasing soil carbon. I have not seen here data that I think supports your claims that enough CO2 can be sequestered in the soil to substantially lower temperatures. The amount you claim can be removed has to be compared to the amount that needs to be removed. The latter has not been addressed.
When you say you do not know how much CO2 can potentially come out of the ocean it appears that you need to do more background research to support your very strong claims. The amount of CO2 involved is potentially many times more than is currently in the atmosphere.
I agree Michael The ocean is the unknown to me. All I have claimed ever is that we can sequester into the soil many times more than all the biomass and atmospheric carbon combined based on the fact that since human impact many times more was lost. Currently right now world wide we lose so much soil there is more abandoned cropland than arable cropland. I could go on and on but this explains it better. Soil Erosion Threatens Food Production
So honestly there is a strong case for rebuilding soil rapidly like the new research in the LCP allows us to do, whether or not it changes AGW or not. But the numbers are larger that all the other climate scientists mitigation proposals combined. More than zero carbon emissions, more than CCS, more than all of them. So either every CO2 mitigation plan ever made is completely useless and nothing will work, or this will work. Simple as that. I don't know if it is large enough to capture all the outgassing of the ocean. But if it can't, then nothing can in under millions of years and nothing we do including 0 emissions has any chances of reversing AGW. I don't believe that any more than I believe AGW denialism. The only difference being 1 denies we did it, and the other denies we can fix it. But both are in complete denial of human impact on the biosphere, whether positive or negative.
Thanks, RB. Very good point about eroded carbon. Has anybody done any studies on the carbon content of recent lake/reservoir sediments downstream from farmed areas?
Red Baroon,
We agree that increasing the carbon in the soil would be a good thing to do. We will have to see how much it helps reduce the carbon burden in the atmpsphere. Every little bit helps some! If it also increases soil productivity (which as a gardener it helps my soil a lot) that is great too.
Michael,
What you continue to ignore is the scale. If the nearly 1.5 billion ha of world cropland now under cultivation for crop production (and a significant % of the 2 billion ha of cropland that has been abandoned by humans since farming began due to soil degradation) were sequestering CO2 at 5 -20 tCO2/ha/yr via the LCP, that totals 7.5 to 30 GtCO2/yr cropland + whatever degraded abandoned land can be restored.
+ 3.5 billion ha agricultural grazing land = 17.5 to 70 Gt CO2/yr
So just those two categories alone we have a biological potential of 25 to 100 GtCO2/yr. And not for only a year or two. The source I already gave you states currently about 80% of the world’s agricultural land suffers moderate to severe degradation. So instead of the 6, 8, 10% SOC meters deep; we have 1, 2, 3% SOC centimeters deep. It will take decades to fully restore this land. That means at MINIMUM we can have 25–100 Gt CO2e pumping into the soil sink for decades before we even have to start worrying about feedbacks slowing us down. In fact no one really knows if it ever will slow down short of atmospheric CO2 getting so low and climate so cold it reduces the flow. All the inovators are claiming sequestration rates that continue to climb. Gabe Brown has one field up to 11%. That field actually sequesters faster now! It just keeps getting deeper. Coli Seis's best fields are up to sequestering 33t/ha/yr. No reduction in rate at all. Dr. Jones asks why would anyone think the LCP would ever slow down?
The soil gets so healthy it kills off the AMF? Maybe so. But no one ever observed such a thing ever.
Total worldwide CO2 emissions from fossil fuels and cement combined was 36 Gt CO2 in 2014, but only about 20Gt actually adds to the atmosphere, because natural ecosystems and processes are at work mitigating this already.
Of course biological potential varies quite significantly from practical potential. Just training all the farmers how to do it would be a herculean task.
That's why I have consistently used far more conservative numbers. I get quite annoyed when I use ultra conservative numbers and get accused of exaggerating them instead, implication being this is some small thing that maybe might help just a little. You claim there is an immense amount of carbon that needs to be sequestered. I agree. However, the land surface of the planet under agriculture is immense! And the amount of carbon missing from our agricultural soils worldwide is immense.
I have been using 62-250% as my numbers. But as you can see I am being excruciatingly conservative.
Red Baron,
Good luck with your scheme. I hope that you can rebuild human agricultural methods across the entire world and that drought does not block your scheme. I will support your enterprise because I think better soil makes for better farming.
I note that you do not cite a single reference to support your claims in 31. Of the very few citations you have previously made most appear to me to be anecdotal or very small scale.
While you claim that large amounts of carbon can be sequestered, you have not addressed how much carbon has to be removed from the atmosphere. You do not account for where all the soil carbon that has been lost has gone. It stands to reason that you cannot sequester all the carbon released from soil and all the fossil carbon by your scheme. You state in 27 that you have no idea how much carbon the ocean will outgas. These issues have been studied and peer reviewed papers address these points. I think you underestimate the amount of carbon that has to be removed by a very large amount. Remember the denominator.
Everyone has their own standard of proof. While I hope you succeed, you have not met my standard. Based on the data I have seen you present, I doubt your claims.
Since I have also offered no data or proof I expect readers to treat my claims with caution. Look at the posts that Tom Curtis makes. He rarely makes a claim without a citation to show why he claims that. When he makes the same claim again he cites his reference again. To convince scientists you need to show that you are correct using data from peer reviewed sources.
That's the problem Michael. Very little has been done on negative emissions scenario's. There is this: Simulating the Earth system response to negative emissions But it explicitly states that
This is not my field. In fact there is no scientist on the planet that can fully have expertise in every field of climate science. It is too complex. I can add my knowlege you can add your and Tom can add his. Everybody from agricultural science to marine biologist, to physicists, mathematicians, economists, geologists etc etc.. Someone has to take the new information available regarding the LCP and add it to their specialty! I can't stop my research go back to school for 6 or 8 years to specialise on modeling a negative emissions scenario, then go back to my research again. The renassance man that knows everything in every field went extinct centuries ago. What you are asking me to do is unreasonable. Nor can Hansen be expected to know the cutting edge of agricultural sciences regarding the LCP. It just isn't reasonable on something this complex. Now I have many places here given case studies and published papers as references. And there are people with different fields of expertise going over them. Read #17 if you don't believe me.
But try as I might, I have yet to find the paper that modeled the scenaio where fossil fuel emissions drops to near zero AND worldwide agriculture changes are made sequestering 25 to 100 GtCO2/yr , what the outgassing of the oceans would be. It just isn't on any of the models on any published paper I can find. And I lack the expertise to publish a paper with than scenario myself.
Wow, I'm impressed with the responses and tangentially inspired responses to my initial inquiry. I don't know if any of you who've posted your learned responses are actually "climate scientists", so if any of you are I'd love to have you say so. I'm just a humble engineer and inventor of a sequestration technology, but one of my friends is a maverick climate scientist from the UK who is not a global warming denier (only a fool would be so), but he is an AGW skeptic. I invited him to create an account with this group but I don't know if he has. His imputs would liven the discussions for sure. I think perhaps I might not have made myself fully clear in my initial inquiry to this group. I did not mean to imply that there isn't an evaporative transfer of heat upwards as precipitation enters the oceans. It wasn't heat per se I was asking about, rather the capture of CO2 in precipitation, and whether or not greater precipitation brought on by atmopsheric warming would capture more CO2 to then be transferred to the oceans. Was this clear to anyone else?
Jeffrey,
Yes I got that was the feedback you were refering to. No I can't comment on it as I am not a climate scientist. Like you I amd working on mitigation through sequestration, in my case agricultural soils. I have taken a climate science course at university level in order to better understand and communicate with climate scientists and better communicate climate science to laymen. But that does not make me one myself. Just a research farmer concerned as all farmers are about climatic conditions and their impacts.
Thanks RedBaron. I love your descriptions of what you're doing. This soil sequestration is something I've wondered about for a long time. What I still wonder about is the eventual surrender of sequestered CO2 back to the atmosphere. Decaying organic matter yields CO2, right? I even have the same concerns for my sequestration technology. My technology captures 95%+ of all gases of combustion but somewhere down the road through various natural processes I'm sure the sequestered CO2 will find its way back into the environment in one form or another. By the way, in my post #34 above I actually know how to spell "inputs". That's a dumb typo.
Jeffrey,
Yes decaying organic matter yields CO2 (and/or CH4 in anaerobic conditions). This is well described by the Roth C mathematical model used by most climate scientists. Only a tiny % of that carbon actually gets sequestered long term in the soil. The rest eventually returns to the atmosphere. I want to emphasize that this model is well developed and I have no problem with it at all in describing the fate of carbon that follows this biological pathway.
Where the difference lies is that it does not describe the newly discovered Liquid Carbon biological Pathway at all. This carbon is not easily decayed. Once that carbon reaches the humic polymer stage, it tightly bonds to the soil mineral substrate and becomes an intrinsic part of the soil matrix. But not only that, it actually stabilizes the soil too.
Remember, these two pathways are fundamentally different. Catabolic pathways like described by the Roth C model break down molecules and produce energy. Anabolic pathways like the LCP synthesize molecules and require energy. That's the primary reason for reducing or eliminating herbicides and other biocides. The energy comes from increased photosynthesis and through the LCP (anabolic pathway) stable soil structure is built. Entirely the opposite of the decay of organic material on the surface. Keep in mind, this is so completely the opposite of what was thought prior to the discovery of Glomalin in 1996, that most soil science textbooks still claim that mollic epipedons are primarily formed by decaying dead roots. And they had no explanation at all why this should happen under grasslands rather than forests which contain more biomass. Keep in mind all this is still contentious. Even this paper published as recently as 2011 and stating the current models are flawed, still describes as "unknown" the reason why. I can't stress enough how exciting this new breakthrough is for both agriculture and climate science.
Now you make a good point questioning the time it can remain sequestered. Once those humic polymers tightly bind to the soil forming a mollic epipedon, this will last into geological time frames unless disturbed. However, we certainly know how to release it if needed. ;)
Thanks, RedBaron. Exciting stuff for sure. Very enlightening.
@34
Jeffrey,
This paper by Turk, et al., touches some of your questions about rain's role in transfering carbon dioxide to the oceans.
"Rain induces surface layer chemical dilution, enhances the gas transfer velocity and exports carbon from the atmosphere by wet deposition."
www.aoml.noaa.gov/phod/docs/2010_Turk_etal_GRL.pdf
Though several factors are addressed, the gist appears to be that where there is little existing wind-driven surface turbulence (such as portions of the tropics), rain creates more surface mixing and can turn a net CO2 source into a net CO2 sink.
"Rain may have a significant effect on air‐sea CO2 exchange directly through increased transfer velocity and indirectly by chemical dilution and wet deposition."
So a final answer would probably involve a more detailed analysis of where climate change is moving precipitation; that is, if climate patterns shifted precipitation away from the tropics and toward more volatile oceans, you would see less CO2 transfer. If climate change simply created more rain over, say, the Southern Ocean, there would likely be a minimal effect.
[PS] Fixed link. In future please create link with the link tool in the comment editor.
Thanks, Jeffronicus. I read through the linked report you posted and it does not answer my initial question posted. As we know CO2 is "captured" by atmospheric water into the molecular form of carbonic acid. Rain has never been "pure", it's always been a weak acidic solution, whether carbonic acid or sulfuric acid. It's virtually impossible for rain to fall upon land or sea as pure H2O. So as global warming increases the amount of evaporated water in the atmosphere, that increased atmospheric water has to capture more CO2, it's unavoidable. And then that increased CO2 capture is going to transport more CO2 to the oceans, either directly through rain falling on the oceans or by way of rivers.